The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.Ĭompeting interests: The authors have declared that no competing interests exist. txt file.įunding: This work was funded by the NIH GM 123019 and NSF DMR 1609784. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.ĭata Availability: The data underlying the results presented in the study have been added as a supplemental. Received: JanuAccepted: JPublished: October 7, 2019Ĭopyright: © 2019 Parr et al. Asthagiri, Northeastern University, UNITED STATES The model makes specific prediction regarding dendritic cell motion and defines conditions for the design of extracorporeal devices for focusing the position of dendritic cells.Ĭitation: Parr A, Anderson NR, Hammer DA (2019) A simulation of the random and directed motion of dendritic cells in chemokine fields. The model also identifies conditions in which dendritic cells can be focused at a single location when confronted with countergradients of chemokine. The model can explain directional motion in chemotactic gradients, as well as the effect of superposition of coincident gradients of chemokine on chemotaxis. We developed a model of dendritic cell motion based on a force balance, in which pulling from filopodia (calculated from a well-established model of filopodial adhesion-clutch dynamics) is counterbalanced by adhesive friction, and where the angular orientation of filopodia is based on the strength of chemotactic signal. 
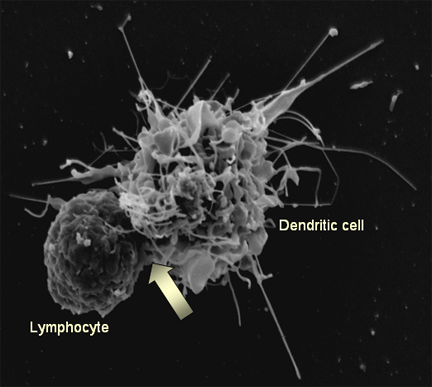
Dendritic cells use filopodia to pull themselves forward, and orient their filopodia based on signals received from chemokines. 
These differences emerge despite both chemokines having virtually the same K d, suggesting a mechanism of signal amplification in DCs requiring further study.ĭendritic cells harvest and display antigen to other immune cells, and motility is essential to their function. Assigning the signal of CCL19 binding CCR7 a baseline strength of 1, we found CCL21 binding CCR7 had a strength of 0.28, and CXCL12 binding CXCR4 had a strength of 0.30. Comparing our simulations to experiment, we can give a quantitative measure of the strength of certain chemokines relative to others. Moreover, gradients are found to be most potent when cells are in a gradient of chemokine whose mean concentration is close to the binding of the K d to the receptor, and least potent when the mean concentration is 0.1K d. We calculated the effect of varying gradient steepness on the line of equistimulation, with steeper gradients resulting in tighter clustering. We predicted that when placed in two opposing gradients, cells will cluster in a line, which we call the “line of equistimulation ” this clustering has also been observed. Cells undergoing chemotaxis exhibited a stronger chemotactic response when exposed to lower average chemokine concentrations, also consistent with experiment. min -1 and a persistence time of 3.2 ± 0.46 min, consistent with experiment.Simulated DCs undergoing chemokinesis were measured to have a speed of 5.1 ± 0.07 μm Our model simulates chemokinesis and chemotaxis in a variety of chemical and mechanical environments. The direction of fliopodial extension is coupled to differential occupancy of cognate chemokine receptors across the cell. In this paper, we simulate the motility of DCs by modeling the generation of forces by filopodia and a force balance on the cell. Despite interest in how DCs respond to chemical stimuli, there have been few attempts to model DC migration. They ferry antigen from the extremities to T cells and are essential for the initiation of an adaptive immune response. Dendritic cells (DCs) are the most effective professional antigen-presenting cell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
